 
They are only lower in energy when they are empty.) The #4s# is higher in energy, not lower, when the orbitals are filled. 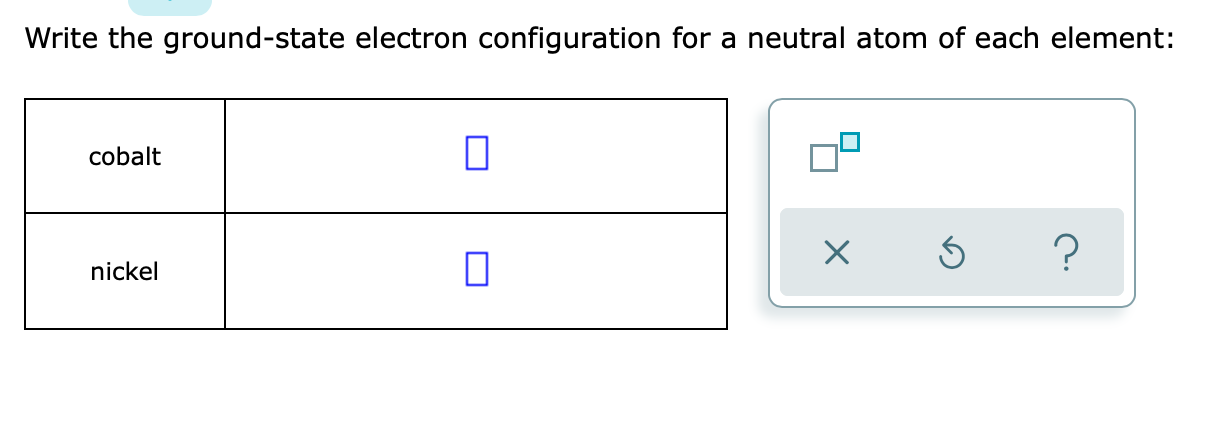
Note that the total number of electrons in the neutral atom adds up to the atomic number, so #2+2+6+2+6 = 18#, which is the atomic number of #"Ar"#.įinally, the #4s# orbitals are higher in energy than the #3d# orbitals by about #"3.75 eV"# when they are partially filled like this, so we write the #3d# before the #4s#. Nevertheless, check the complete configuration and other interesting facts about Cobalt that most people dont know. To write the configuration for the Cobalt ions, first we need to write the electron configuration for just Cobalt (Co). Writing the electron configuration, you really only need the valence orbitals, and you can omit the core orbitals by notating it via the noble gas shortcut. (Bi) Bismuth is a chemical element of the periodic table with chemical symbol Bi and atomic number 83 with an atomic weight of 208.98 u and is classed as post-transition metal and is part of group 15 (nitrogen group). In the case of Cobalt the abbreviated electron configuration is Ar 3d7 4s2. Electronic configuration of the Radium atom in ascending order of orbital energies: 2020-2023 Your online. Ra (Radium) is an element with position number in the periodic table. Its valence orbitals are the #4s# and #3d#'s. Electron configuration for Radium (element 88).If your periodic table doesn’t agree with this, your answers for elements near the f-orbitals may be slightly different. Its core orbitals are the #1s#, #2s#, #2p#'s, #3s#, and #3p#'s. Note: The electron configurations in this worksheet assume that lanthanum (La) is the first element in the 4f block and that actinium (Ac) is the first element in the 5f block.Hassium: Value is a guess based on periodic. Bohrium: Value is a guess based on periodic table trend. Seaborgium: Value is a guess based on periodic table trend. 
The oxidation state of the element changes depending on the bond formation. Notes on the Electron Configuration of particular elements: Dubnium: Value is a guess based on periodic table trend. Bromine atom exhibit -1, +1, +3, +5 oxidation states. The electron configuration of bromide ion(Br ) shows that the bromide ion acquired the electron configuration of krypton. Iron is on the fourth row of the periodic table, sixth column of the transition metals, atomic number #26#. The electron configuration of bromide ion(Br ) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
